 
There is no hard and fast rule for this, but that is an explanation that correlates with experimental data. Cobalt has 1 stable naturally occuring isotopes while Chromium has 4 stable naturally occuring isotopes. Thus, electron pairing is favorable enough for Tungsten. The more the electron distribution is spread out, the less electron-pair repulsion there is, and thus the lower #Pi_c# is. However, Tungsten's #5d# and #6s# orbitals being larger than the #3d# and #4s# orbitals (respectively) spreads out the electron density enough that the pairing energy ( #Pi = Pi_c + Pi_e#) is small enough. The small-enough orbital size means that the electron density is not as spread out as it could be, which makes it favorable enough for a maximum total spin to give the most stable configuration.The minimization comes from having all unpaired electrons in the #3d# and #4s# ( #3d^5 4s^1#), rather than one electron pair in the #4s# ( #3d^4 4s^2#). The minimized coulombic repulsion energy #Pi_c# further stabilizes this configuration.The maximization comes from how there are #5# unpaired electrons, instead of just #4# ( #3d^4 4s^2#). The maximized exchange energy #Pi_e# stabilizes this configuration ( #3d^5 4s^1#).One explanation for Chromium, then, is that: The former is stabilizing and the latter is destabilizing, as shown below (suppose configuration 2 is at pairing energy #Pi = 0#): These combine to produce an overall pairing energy #Pi = Pi_c + Pi_e#.Louis Nicolas Vauquelin first discovered chromium in 1797 and first isolated it the following year. The number of electrons in each of Chromiums shells is 2, 8, 13, 1 and its electron configuration is Ar 3d 5 4s 1. The coulombic repulsion energy #Pi_c# (a destabilizing factor that is inversely proportional to the number of electron pairs) Chromium (atomic symbol: Cr, atomic number: 24) is a Block D, Group 6, Period 4 element with an atomic weight of 51.9961.The exchange energy #Pi_e# (a stabilizing quantum mechanical factor that is directly proportional to the number of pairs of electrons in the same subshell or very close-energy subshells with parallel spins) But chloride ion formed (when chlorine accepts an electron) has noble gas configuration of argon. English: An SVG periodic table of the elements, which includes name, atomic mass, electron configuration, first ionization energy, and electronegativity. 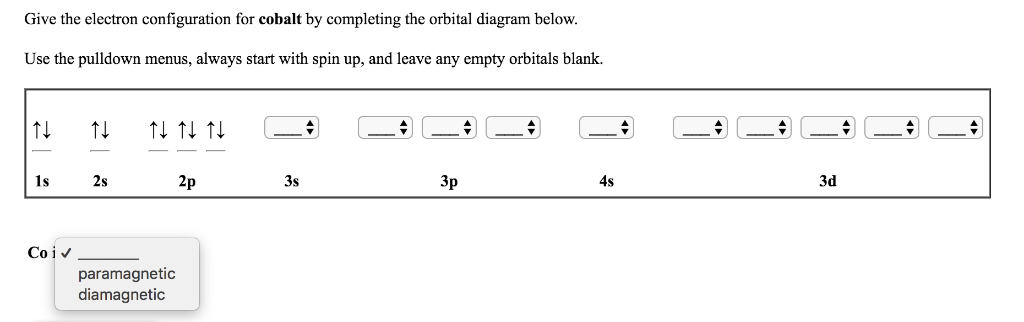
To explain Chromium's electron configuration, we could introduce: 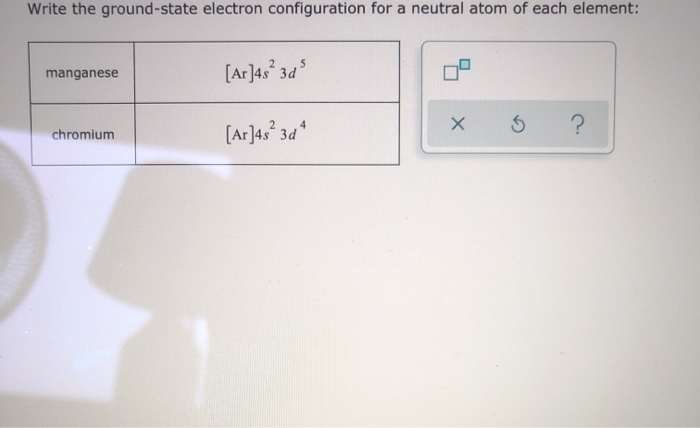
Unfortunately, there is no easy way to explain these deviations in the ideal order for each element. Interestingly enough, Tungsten is more stable with an electron arrangement of #4f^14 5d^4 6s^2#. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
