 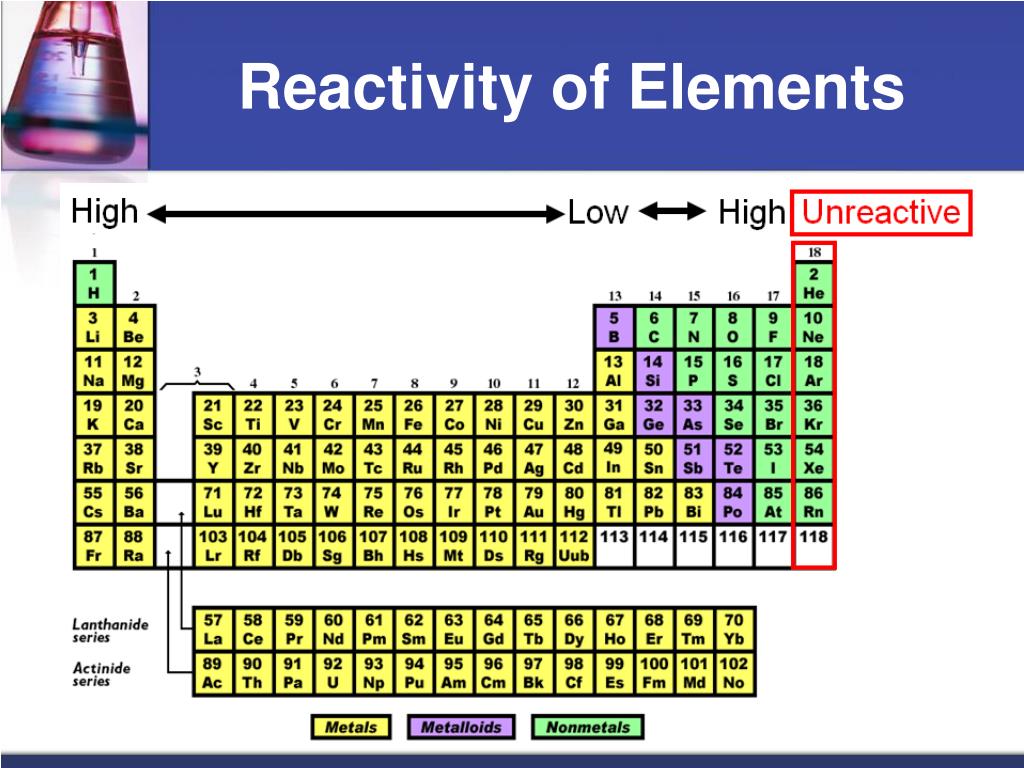
According to this criterion, pyridine is more aromatic than furan, but it is difficult to say by how much. As a result, the degree of aromaticity is measured in terms of reactivity by the relative tendency toward substitution rather than addition. The extra stability, in turn, determines the compound’s tendency to react by substitution of hydrogen-that is, replacement of a singly bonded hydrogen atom with another singly bonded atom or group-rather than the addition of one or more atoms to the molecule via the breaking of a double bond (see substitution reaction addition reaction). The extra stability of the conjugated system that an aromatic compound contains affects its reactivity. Chemical reactivity can provide qualitative information about aromaticity. The periodic trends allow for a partial inference of any element’s unknown properties There are a few exceptions, such as the ionization energy trend of group 3, the electron group 17 elements’ affinity trend, group 1 elements’ density trend (alkali metals), and so on. Based on their atomic structures and properties, these laws allow chemical elements to be organized in the periodic table. Changes in the atomic structure of the chemical elements within their respective periods (horizontal rows) and groups (vertical columns) in the periodic table cause periodic trends. Electronegativity, ionization energy, electron affinity, atomic radii, ionic radius, metallic character, and chemical reactivity are all major periodic trends. Periodic trends are specific patterns in chemical element properties revealed by the periodic table of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
